EMA
MAH
June 2, 2022
EMA: Procedural advice for vaccine platform technology master 5 file (vPTMF) certification (DRAFT)
This document is intended to provide advice to marketing authorisation (MA) applicants and marketing authorisation holders (MAHs) on issues associated with the submission, evaluation, certification...
EMA
MAH

May 24, 2022
EMA post-authorisation procedural advice for users of the centralised procedure
This guidance document addresses a number of questions which marketing authorisation holders (MAHs) may have on post-authorisation procedures. It provides an overview of the Agency’s position on...
EMA
Europe

May 20, 2022
IRIS guide for applicants (How to create and submit scientific applications, for industry and individual applicants)
This guide has been produced to show applicants how to use the IRIS platform to prepare and submit an application and/or data for a scientific procedure (orphan designation application, scientific...
EMA
Europe

May 19, 2022
EMA Regulatory and procedural guideline: Public consultation concerning the physical attendance and the location of personal residency of the qualified person
The COVID-19 pandemic required manufacturers and importers of medicinal products and regulatory authorities to operate under business continuity mode, impacting the standard way of working. As a...
EMA
Europe

April 26, 2022
EMA and the EUnetHTA 21 consortium set priorities for their collaboration Share
April 12, 2022 The European Medicines Agency (EMA) and the European Network for Health Technology Assessment (EUnetHTA) 21 consortium have published a PDF icon joint work plan until 2023. The focus...
EMA
EU
![]()
April 26, 2022
Facilitating global access to diabetes treatments for non-EU patients Share
April 22, 2022 EMA’s human medicines committee (CHMP) has given a recommendation for two diabetes mellitus treatments, Actrapid and Insulatard, for use outside the European Union (EU). EMA is...
FDA
EMA
![]()
February 4, 2022
FDA’s Breakthrough Therapy Designation vs PRIority MEdicines (PRIME) Application in Europe
What are Breakthrough Therapy Designation and PRIority MEdicines (PRIME) Applications? The advancement of modern medicine, and the accessibility of researched and regulated medication, has greatly...
EMA
EU
![]()
December 22, 2021
Regulatory Strategy for Clinical Trials in the European Union
Regulatory Strategy for Clinical Trials in the European Union: Setting up a clinical trial in the European Union (EU) has historically been an expensive and time-consuming business. With 27...
EMA
ICH
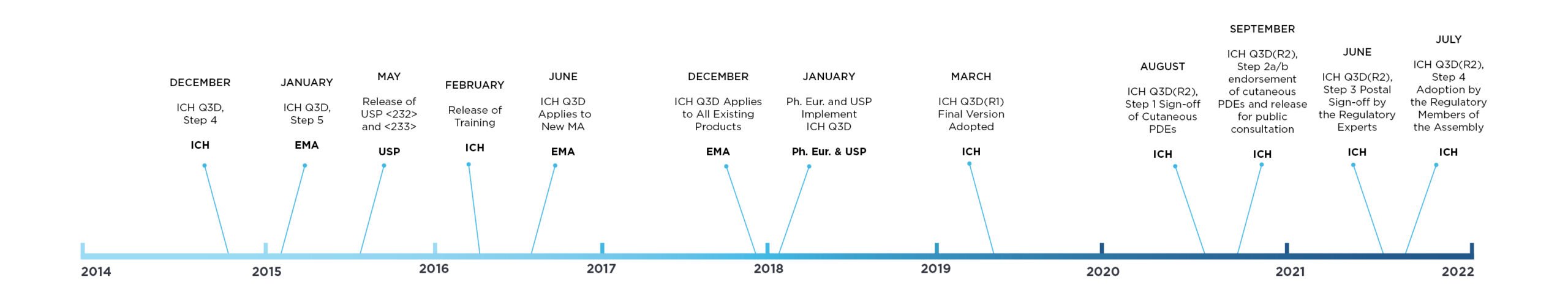
March 11, 2021
How to Manage the Risk of Elemental Impurities with ICH Q3D
How to Manage the Risk of Elemental Impurities with ICH Q3D: The mission of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) is to ensure...






