Clinical Research Solutions
Four Benefits of Requesting an FDA Pre-IND Meeting
Although not required, a Pre-IND Meeting is a critical milestone, one that is highly recommended by the FDA. The goal of the meeting is to receive general agreement from FDA that your drug...
Clinical Research Solutions
EMA Takes Steps to Minimize Animal Testing During Product Development
On Wednesday, September 29th, EMA announced the implementation of new measures indented to replace, reduce, and refine animal use in the development, manufacturing, and testing of human and...
Clinical Research Solutions
Critical Issues in Drug Development: Don't Get Derailed in Developmental Efforts
Pharmaceutical companies spend massive amounts of money on drug development. Regardless of a firm’s size, the investment is substantial; the smallest error can have catastrophic consequences,...
Clinical Research Solutions
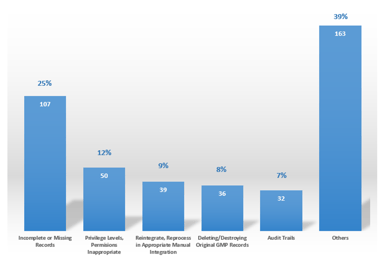
Five Steps Toward a Mature Data Integrity Culture
The corporate and quality culture has a significant effect on the maturity level of Data Integrity within a regulated company and should, therefore, be assessed and understood. To achieve the level...
Clinical Research Solutions
How to Present Scientific Data That Generates Regulatory Approvals
Today’s patients benefit from having access to the safest, most advanced pharmaceutical systems around the world. This is because of the work done by the various regulatory bodies who govern the...
6 Benefits of Custom PMO Design
A Project Management Office ("PMO") can be incredibly advantageous to large companies with many ongoing projects across various job sites. A PMO streamlines project management processes to make a...
Clinical Research Solutions
Life Science Outsourcing: Not sure you’re ready to turn over the keys?
It's no secret. To be successful in the life sciences industry you must accomplish a lot – achieve and maintain profitable growth, deliver safe and effective products to patients quickly, manage...
Clinical Research Solutions
Understanding The Medical Device Single Audit Program
The Medical Device Single Audit Program (MDSAP), initiated by the International Medical Device Regulators Forum (IMDRF), created a global approach to auditing and monitoring the manufacturing of...
Clinical Research Solutions
The Human Factor - Preparing Your Device for Usability Testing
When it comes to usability studies, the focus should be on effectively preparing the medical device for use in humans. Whether a Sponsor is conducting formative testing or validation testing, the...

FDA Guidance on Gene Therapy Products: RCR Testing and Monitoring – Focus on Documenting Test Results & Post-Licensure Considerations
In January 2020, FDA issued a final guidance document on the topic of "Testing of Retroviral Vector-Based Human Gene Therapy Products for Replication Competent Retrovirus During Product Manufacture...
Clinical Research Solutions
Fundamentals of Data Integrity for ATMP Development
Advanced Therapy Medicinal Products (ATMPs), or Cell and Gene Therapies (CGT), have the incredible potential to cure devastating illnesses, such as cancer, on a more personalized level. But, due to...
Pedaling While Fixing a Flat: Conducting a Good FDA Meeting Under Bad Circumstances
Before using a significant risk medical device in a clinical study, Sponsors must first obtain FDA approval. Frequently, the request for an investigational device exemption (IDE) is disapproved, and...
Clinical Research Solutions
45 Years of 510(k) Submissions: FDA Celebrates with Improvements to the Process
In late 2018, FDA announced changes to modernize and increase the efficiency of its 510(k) clearance pathway, which is the most common medical device submission and allows for the allows for the...
Clinical Research Solutions
Understanding Cybersecurity Threats to Medical Devices
The threat of cyber-attacks against medical devices is real. Medical devices capable of connecting, wirelessly, wired, or to portable media such as a USB drive, are more vulnerable to cybersecurity...

How to File Post-Approval Changes to an NDA or ANDA
On November 21, 1997, the Food & Drug Modernization Act (the Modernization Act) was enacted by Congress. Section 116 of the Modernization Act amended the Federal Food, Drug, and Cosmetic (FD&C) Act...
Clinical Research Solutions
What to Expect From an FDA Inspection, Part 2
In this continuation from “Preparing for an FDA Inspection” I will discuss what I have witnessed as a typical outline of events for FDA inspections. Prior to setting foot at the facility, the FDA...