Why You Need a Decentralized Clinical Trials Strategy - Before Disaster Strikes
Imagine having your clinical trial ready to go, or already underway, and overnight the brakes are slammed, bringing your trial to a screeching halt. This was the reality for sponsors around the world...
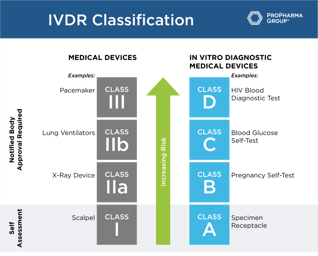
What the IVDR Is and How to Prepare
In May 2022, the IVDD will be repealed by the European Committee, thereby ending the transition period. To ensure you're compliant with IVDR by that date, learn everything you need to know about the...
Battle of the Regulators: Comparing FDA’s Accelerated Approval and EMA’s Conditional Marketing Authorisation
The Food and Drug Administration (FDA) and the European Medicines Agency (EMA) offer various pathways that can expedite the development and review of new therapies to treat serious or...
Clinical Research Solutions
Roadmap for Successful IVDR Transition, Part II: Technical Documentation & Software
The compliance dates for the In Vitro Diagnostics Regulation (IVDR) are quickly approaching (May 2022). In this blog series, we discuss the final months before the IVDR date of application along with...
Meet the Expert: Marshall Scicchitano
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...
Clinical Research Solutions
FDA’s Breakthrough Therapy Designation vs PRIority MEdicines (PRIME) Application in Europe
What are Breakthrough Therapy Designation and PRIority MEdicines (PRIME) Applications? The advancement of modern medicine, and the accessibility of researched and regulated medication, has greatly...
Clinical Research Solutions
Roadmap for Successful IVDR Transition
Roadmap for Successful IVDR Transition: The compliance dates for the In Vitro Diagnostics Regulation (IVDR) will become effective on May 26, 2022. To help you with the IVDD to IVDR transition, we've...
Clinical Research Solutions
Top Questions & Answers for Managing the New IVDR
With an increased need for high quality in vitro diagnostic medical devices (IVDs), the In Vitro Diagnostic Regulation (IVDR, 2017/746) was entered into force for all IVDs in 2017 with a five-year...
Should You Undergo Virtual Factory Acceptance Testing?
Although domestic travel is slowly ramping up, companies may still consider virtual factory acceptance testing (FAT). FAT is an optional step in the life sciences’ process of purchasing factory...
Regulatory Sciences
Regulatory Strategy for Clinical Trials in the European Union
Setting up a clinical trial in the European Union (EU) has historically been an expensive and time-consuming business. With 27 individual member states each requiring its own review and approval, it...
Regulatory Sciences
Is it Time to Sharpen Your Target Product Profile (TPP)?
Is it Time to Sharpen Your Target Product Profile (TPP)? We’ve heard it countless times: "Fail early and live to fight another day." "Preserve capital, human resources, and energy for a project with...
Clinical Research Solutions
7 Benefits of Working with a Biostatistician
Biostatisticians have become increasingly important as many data-driven public health projects and clinical trials have expanded. So, what exactly does a biostatistician do? These individuals are...
A Roadmap for Clinical Trials: QP Certification of IMP
Please note that updates to regulations may have been implemented since the publication of this article. The 20th century was the century of many scientific discoveries in the field of medicine....
Clinical Research Solutions
Understanding Bioequivalence and Product-Specific Guidances
The FDA regularly issues new and revised product-specific guidances to facilitate the availability of generic drugs and assist the generic pharmaceutical industry with identifying the most...

Regulatory Sciences
European Expedited Regulatory Programs Explained
Do you really know how to accelerate the approval of your innovative product in Europe? The FDA's incentives for promising new medicines are widely known. Accelerated approval, priority review, fast...
Meet the Expert: Maurice Weijers
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...