
Clinical Research Solutions
UK Paediatric Investigational Plans – what do you need to know?? …and how is it all working in practice??
If a marketing authorisation is planned to be submitted in England, Scotland, and Wales (GB), an MHRA-approved paediatric investigational plan (PIP) is required. Up until January 1, 2021, PIPs were...
How to Fast-Track medicine approval in the UK with the MHRA’s Innovative Licensing and Access Pathway (ILAP)
What is ILAP? What benefits does ILAP provide? How do you access it? With the dust of Brexit settling, the question on most people’s lips (well, those of us in the healthcare sector anyway!) was:...

Clinical Research Solutions
5 Benefits of Receiving EU PRIME Designation for Medicine Developers
What it is, why it matters, how you can apply and how we can help. What is the PRIME Scheme? You might be forgiven if you don’t know what the PRIority MEdicines (PRIME) scheme is; but if you are in...
Orphan Drug Designations in the US and EU
What is an Orphan Drug Designation? The Orphan Drug Designation (ODD) program in both the United States (U.S.) and European Union (E.U.) qualifies sponsors to receive potential incentives to develop...
Clinical Research Solutions
Global regulators agree on key principles on adapting vaccines to tackle virus variants
On 30 June, regulators from around the world discussed emerging evidence to support adaptation of COVID-19 vaccines as the SARS-COV-2 virus continues to evolve during a workshop co-chaired by the...
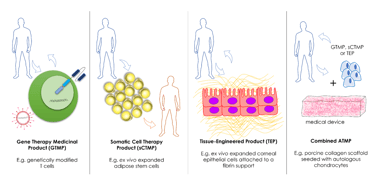
How to Leverage EMA’s ATMP Classification
Advanced therapy medicinal products (ATMPs) have emerged as ground-breaking therapies for rare diseases and other conditions with unmet clinical needs. As of 2022, sixteen ATMPs have been approved by...

Clinical Research Solutions
The 5 Phases of Project Management + Pro Tips for Success
Guilty as charged! I used to think a project manager’s (PM) sole job was to remind everyone about deadlines and set up status meetings. I learned firsthand while working at a medical device start-up,...
Meet the Expert: Jennifer Daudelin
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...
Maximising on Scientific Advice Procedures in Europe
A unique opportunity to interact with medicine regulators in Europe Are you considering requesting scientific advice in Europe? We can help you navigate the various procedures within the European...
EMA adopts first list of critical medicines for COVID-19
News On 7 June 2022, EMA's Medicines Shortages Steering Group (MSSG) adopted the list of critical medicines for the COVID-19 public health emergency. The medicines included in the list are authorised...
Meet the Expert: Elina Koli
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...

Quality & Compliance
What is Product Lifecycle Management and Why is it Critical for Success?
Product Lifecycle Management (PLM) is the process of managing a product from conception through end of of life (EOL), and clearly includes conception (design and development) and commercialization....

Understanding ICH M10 “Bioanalytical Method Validation and Study Sample Analysis"
The final version of ICH M10 “Bioanalytical Method Validation and Study Sample Analysis” was adopted on May 24, 2022. This is the harmonized guideline which has been ratified by participating...

Clinical Research Solutions
Compounded drug search added to FDA’s NDC directory webpage
FDA recently added a search function to the National Drug Code (NDC) Directory webpage for human drugs compounded by outsourcing facilities that assign NDC numbers to their products. This update was...
Meet the Expert: Markus Ganzlin
Our “Meet the Expert” series introduces you to our team of experts around the world. This “behind the curtain” view will help you get to know who we are on a professional and personal level, and...
