Ban on Titanium Dioxide (E171) on the EU Food Market: What Are the Consequences for Medicines?
Use of Titanium Dioxide in the EU Food Market In 2021, the European Food Safety Authority (EFSA) investigated the safety of the white coloring agent titanium dioxide (TiO2) and concluded that the...

Be Careful What You Ask For (Prior to Consent)
According to FDA’s clinical trial regulations (21 CFR 50.20, 312.60 and 812.100), clinical investigators are responsible for protecting the rights, safety, and welfare of subjects during a clinical...

Your Vendor Audit Program: On-site or Remote / Virtual?
It is quite common that a sponsor company will outsource services to external vendors, whether for additional expertise, remote locations, or simply due to lack of availability of resources within...
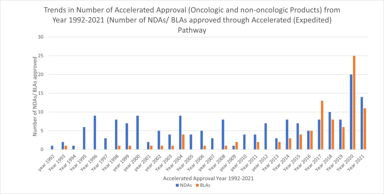
FDA Accelerated Approval Pathway: A Potential Missed Opportunity for Sponsors
The accelerated approval provisions of FDASIA in section 506(c) of the FD&C Act provide that FDA may grant accelerated approval to: . . . a product for a serious or life-threatening disease or...
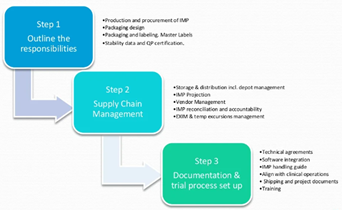
Clinical (IMP) Drug Supply…It’s Complicated
Things to consider and how to ease the process IMP Supply Management is a journey where GMP meets GCP and GDP. This journey includes finance, flow of products and documentation. How a company manages...

What You Need to Know About Developing Vaccines
An unlikely beacon of hope from the otherwise disastrous Covid pandemic, may come in the form of renewed attention towards approaches to vaccine development. The Importance of Vaccines The...

Meet the Expert: Sharon Charles
Our "Meet the Expert" series introduces you to our team of experts around the world. This "behind the curtain" view will help you get to know who we are on a professional and personal level, and...

How to Comply with the Nitrosamine Regulations for Your New Drug Product Marketing Applications
Introduction: Are you in the development phase for your medicinal product? Have you assessed your manufacturing processes with respect to the requirements for investigating the potential presence of...





