Rethinking Biometrics Resourcing in Modern Clinical Trials
Rethinking biometrics as a strategic function to improve consistency, scalability, and clinical trial performance.
Quality & Compliance
At the beginning of each federal fiscal year, the US FDA posts the previous year's Form 483 observation metrics issued by each product center. Inspections ending between 10/1/2017 and 9/30/2018, for...
Who says you can’t teach an old dog a new trick? Having spent the last 25+ years in small molecule, large molecule, medical devices, I have spent a lot of time planning and executing everything from...
ProPharma Group has launched a "Meet the Expert" series to introduce you to our experts from around the world. This series will help you get to know who we are, and how our colleagues work to support...
Regulatory Sciences
Operating a business under a Consent Decree is extremely difficult; there is no area of the business that goes unaffected. In addition to fines that could reach $500 million, a three to five-year...
On Thursday, March 7th, FDA published a revised draft guidance, updating its nonproprietary naming convention for biological products licensed under section 351 of the Public Health Service Act (PHS...
Quality & Compliance
The currently scheduled transition date in the Brexit process, 30 March 2019, is coming very close. In light of this, the European Commission (EC) published on 25 February 2019 a notice on the...
Regulatory Sciences
Earlier this month, FDA announced the approval of Spravato (esketamine) nasal spray. The product is to be used in conjunction with an oral antidepressant and is indicated for the treatment of...

If you check Apple’s App Store or Google’s Play Store you will find an overwhelming list of health and fitness apps. This list only gets longer, if you include the number of people who use these...
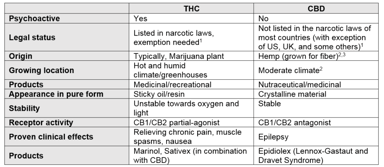
Mention the word cannabis and the confusion starts; legal or illegal, nutraceutical or medicinal product, psychoactive or non-psychoactive, clinically significant or not. At the same time, the...
Regulatory Sciences
On Tuesday, February 5th, FDA published a draft guidance explaining the principles for premarket review of combination products, including how to determine which type of premarket submission is...
Clinical Research Solutions
On Wednesday, January 16th, the FDA published its first draft guidance since the partial government shutdown took effect on December 22, 2018. The draft guidance, entitled "ANDA Submissions –...

Regulatory Sciences
From FDA's approval of the first cannabis-based product in the U.S., to the classification of two Apple Watch apps and changes in the Agency's submission requirements related to Sponsor meetings,...
Each of us makes important decisions that shape our lives, every day. We try to make the best decisions possible and yet, as the adage goes "all predictions of the future are wrong, some are just...
Regulatory Sciences
Over the past 20 years, the traditional approach to drug development has expanded to include the outsourcing of a range of testing and manufacturing functions. As a part of their long-term strategic...

Compiling and submitting a New Drug Application (NDA) submission is a complicated and intensive activity. Once you have submitted your application to FDA, you may be curious about what can you expect...

What’s Next for Rare Disease Innovation? For children suffering from rare and often life-threatening diseases, access to new and effective treatments can mean the difference between life and death....

A Safety Data Exchange Agreement (SDEA) or Pharmacovigilance Agreement (PVA) is a legal written contract which ensures that all safety data, PV tasks and responsibilities are detailed between two or...

What is a PSMF? The EU Pharmacovigilance System Master File (PSMF) is a legally required document created and maintained by a Marketing Authorisation Holder (MAH). Essentially, the document is a...

In order to incentivize the development of therapies (drugs biologics) to fill unmet medical needs for treatment of serious conditions, the FDA has developed various programs to expedite drug...
The FDA’s homepage contains an item in the Public Health Focus section from the Office of Communications entitled “Don’t Use Expired Medicine.” Given this mandate, I recall from many years ago the...