Rethinking Biometrics Resourcing in Modern Clinical Trials
Rethinking biometrics as a strategic function to improve consistency, scalability, and clinical trial performance.

Regulatory Sciences
On Friday, October 12th, FDA issued a guidance document entitled “Impact of Certain Provisions of the Revised Common Rule on FDA-Regulated Clinical Investigations.” The document aims to help...
New rules, old facilities. How do these two meet? Is it a big black hole or is there light at the end of the tunnel? When you work in an older facility, you are probably acquainted with one-liners...

Clinical Research Solutions
On Monday, October 1, FDA’s Center for Biologics Evaluation and Research (CBER) issued a document outlining the policies and procedures for scheduling and conducting INitial Targeted Engagement for...

On Tuesday, September 25th, the FDA published a draft guidance containing recommendations on good review management principles and practices (GRMPs) for new drug applications (NDAs), Biologics...
Regulatory Sciences
On Thursday, September 6th, the FDA released a new draft guidance regarding benefit-risk determinations in medical device premarket approval applications (PMAs), De Novo requests, and humanitarian...
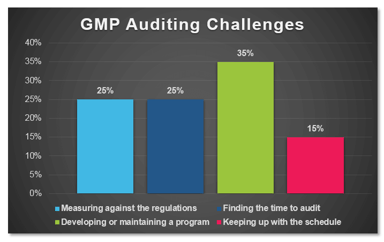
In a recent poll conducted by ProPharma Group, the question “What is your biggest GMP auditing challenge?” was posed to Quality professionals in the drug manufacturing industry. The following graph...
Clinical Research Solutions
Generic drugs are immensely important to the U.S. healthcare system. These drugs account for 89% of the prescriptions dispensed in the United States. And, over the last decade, generic drugs have...

On Wednesday, August 1st, FDA released a notice with updated prescription drug user fee rates for fiscal year 2019. Prescription Drug Application Fees According to the Food, Drug, & Cosmetic (FD&C)...
Quality & Compliance
According to recent FDA updates on the implementation of the Safety and Innovation Act (FDASIA), nearly 40 percent of finished drugs are being imported, and nearly 80 percent of active ingredients,...
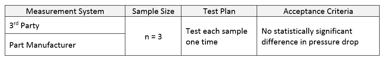
Data collection and analysis is expensive and has the potential to compromise a company’s hard-won compliance position. It is therefore critical that technical leaders follow a systematic and proven...
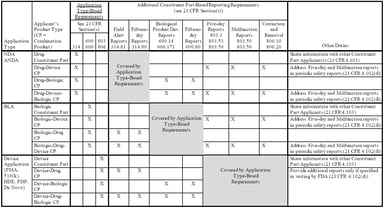
Pharmacovigilance
On March 20, 2018, the US Food and Drug Administration (FDA) released two new guidance documents to help companies comply with the December 20, 2016 final rule establishing postmarketing safety...
MassBio is a non-profit organization that represents and provides support for the life sciences supercluster in Massachusetts. MassBio is committed to growing the industry, adding value to...
Regulatory Sciences
Throughout 2017, the FDA focused its attention on the regulation of generic drug products. In 2015, the Agency issued only two generic-related guidance documents. In 2016, there were seven. In 2017,...
Clinical Research Solutions
Earning FDA approval for your drug program is a journey. A misstep at any point of that journey could jeopardize your chance at getting your drug approved. That’s why it’s so important to have a...
At the beginning of each federal fiscal year, the US FDA posts the previous year’s Form 483 observation metrics issued by each product center. I find that reviewing these metrics provides a valuable...

Compiling and submitting a New Drug Application (NDA) submission is a complicated and intensive activity. Once you have submitted your application to FDA, you may be curious about what can you expect...

What’s Next for Rare Disease Innovation? For children suffering from rare and often life-threatening diseases, access to new and effective treatments can mean the difference between life and death....

A Safety Data Exchange Agreement (SDEA) or Pharmacovigilance Agreement (PVA) is a legal written contract which ensures that all safety data, PV tasks and responsibilities are detailed between two or...

What is a PSMF? The EU Pharmacovigilance System Master File (PSMF) is a legally required document created and maintained by a Marketing Authorisation Holder (MAH). Essentially, the document is a...

In order to incentivize the development of therapies (drugs biologics) to fill unmet medical needs for treatment of serious conditions, the FDA has developed various programs to expedite drug...
The FDA’s homepage contains an item in the Public Health Focus section from the Office of Communications entitled “Don’t Use Expired Medicine.” Given this mandate, I recall from many years ago the...