Clinical Research Solutions
Changes in Medical Device Regulatory Requirements in Europe (2021)
In May 2021, the new European regulations on medical devices (EU MDR) will take full effect. Organizations face a unique set of compliance challenges, mainly due to the numerous changes and additions...

Clinical Research Solutions
The Importance of the eCTD Structure for FDA Approval
The Importance of the eCTD To harmonize the process of regulatory reviews for global drug development, a unified structure was developed and implemented for electronic Common Technical Document...

Clinical Research Solutions
Medical Writing Lean Overview
When applied rigorously and comprehensively, lean principles can reduce organizational costs while increasing productivity and strengthening the overall quality of submissions What is “Lean” as it...

Clinical Research Solutions
6 Unique Challenges Hindering Oncology Clinical Trials
The research for cancer treatment is moving forward at a rapid pace. We're witnessing a shift from chemotherapy protocols to MTAs (molecularly targeted agents) for immunotherapies in particular....
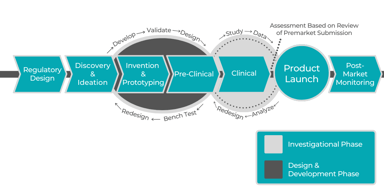
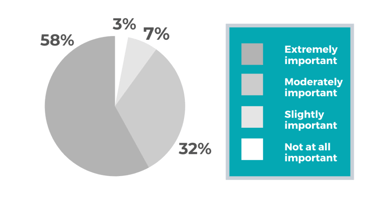
Pharmaceutical Consulting during Product & Business Development
To stay competitive in the pharmaceutical field, pharmaceutical companies need to have access to legal, product and clinical development, business strategy, and human resources experts. Startups...

Clinical Research Solutions
9 Medical Writing Mistakes to Avoid
With around 2.5 million scientific papers published every year, it is impossible for medical professionals to keep up with all the latest research. Every piece of content is fighting for eyes. To...
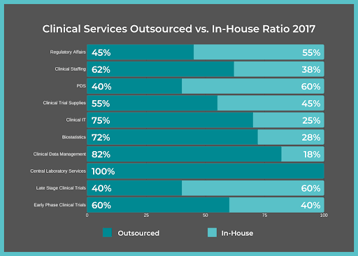
FSP Solutions
5 Ways a Functional Service Provider Can Support Your Clinical Development Project
When pharmaceutical companies launch a clinical trial or reach a certain phase of Clinical Development, with only the support of their in-house employees, the additional workload often becomes too...