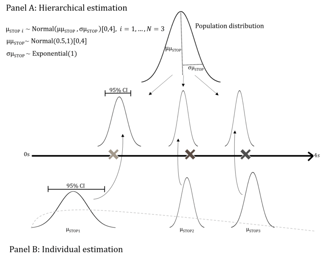
FDA Designates Empirically Based Bayesian Emax Models for Dose Finding as ‘Fit-For-Purpose’
FDA Designates Empirically Based Bayesian Emax Models for Dose Finding as ‘Fit-For-Purpose’: On August 5, 2022, the U.S. Food and Drug Administration (FDA) designated ‘Empirically Based Bayesian Emax...

FDA Issues FY2021 Report on the State of Pharmaceutical Quality
FDA Issues FY2021 Report on the State of Pharmaceutical Quality: The Office of Pharmaceutical Quality (OPQ) within FDA’s Center for Drug Evaluation and Research has published the fiscal year 2021...

FDA Issues Revised Draft Guidance on Charging for Investigational Drugs Under an Investigational New Drug Application
August 2022 Draft Guidance This guidance provides information for industry, researchers, physicians, institutional review boards (IRBs), and patients about the implementation of FDA’s regulations on...
FDA Publishes Responses to Good Clinical Practice Inquiries
FDA Publishes Responses to Good Clinical Practice Inquiries: FDA oversees clinical trials to ensure they are designed, conducted, analyzed and reported according to federal law and FDA’s regulations....

Hydrogen Peroxide-Based Contact Lens Care Products: Consumer Labeling Recommendations - Premarket Notification (510(k)) Submissions
Draft Guidance for Industry and Food and Drug Administration Staff AUGUST 2022 FDA is issuing this draft guidance to provide labeling recommendations for hydrogen peroxide-based contact lens care...

Replacement Reagent and Instrument Family Policy for In Vitro Diagnostic Devices
GUIDANCE DOCUMENT - Guidance for Industry and FDA Staff AUGUST 2022 In 2003, FDA issued an updated guidance on the "Replacement Reagent and Instrument Family Policy" for in vitro diagnostic (IVD)...

FDA Regional Implementation Guide for E2B(R3) Electronic Transmission of Individual Case Safety Reports for Drug and Biological Products
This technical specifications document is to assist interested parties in electronically submitting individual case safety reports (ICSRs) (and ICSR attachments) to the Center for Drug Evaluation and...

EMA: List of centrally authorised products requiring a notification of a change for update of annexes
August 2022 EMA Regulatory and Procedural Guideline Parallel distributors are only required to inform the EMA of changes to the labelling or leaflet related to any update of the annexes of marketing...





