
USP and FDA Propose Updates to Good Storage and Distribution Practices
Updates have been announced by FDA and for USP <1079>. In this blog we cover these changes. USP USP <1079> has a series of chapters on Good Storage and Distribution Practices. Chapter <1079> applies...
FDA Announces Availability of Multiple Draft Guidances - Drug Information Update
Today, FDA announced the availability of the following draft guidances for industry, which were prepared under the support of the International Council for Harmonisation of Technical Requirements for...
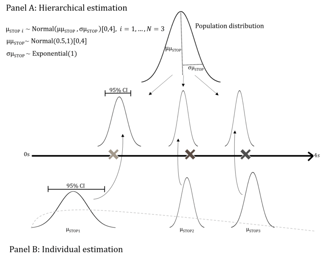
FDA Designates Empirically Based Bayesian Emax Models for Dose Finding as ‘Fit-For-Purpose’
FDA Designates Empirically Based Bayesian Emax Models for Dose Finding as ‘Fit-For-Purpose’: On August 5, 2022, the U.S. Food and Drug Administration (FDA) designated ‘Empirically Based Bayesian Emax...

FDA - Tobacco Products: Principles for Designing and Conducting Tobacco Product Perception and Intention Studies
FDA - Tobacco Products: Principles for Designing and Conducting Tobacco Product Perception and Intention Studies. August 2022 This final guidance is intended to help applicants (or “you”) design and...

FDA Issues FY2021 Report on the State of Pharmaceutical Quality
FDA Issues FY2021 Report on the State of Pharmaceutical Quality: The Office of Pharmaceutical Quality (OPQ) within FDA’s Center for Drug Evaluation and Research has published the fiscal year 2021...

FDA Issues Revised Draft Guidance on Charging for Investigational Drugs Under an Investigational New Drug Application
August 2022 Draft Guidance This guidance provides information for industry, researchers, physicians, institutional review boards (IRBs), and patients about the implementation of FDA’s regulations on...
FDA Publishes Responses to Good Clinical Practice Inquiries
FDA Publishes Responses to Good Clinical Practice Inquiries: FDA oversees clinical trials to ensure they are designed, conducted, analyzed and reported according to federal law and FDA’s regulations....
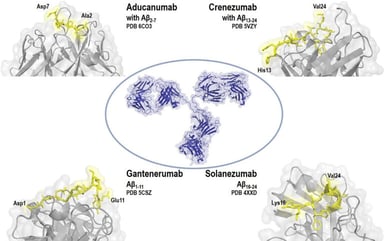
Orphan Designation of ATMPs for Rare Diseases: MPS II Case Study
Orphan Designation of ATMPs for Rare Diseases: MPS II Case Study Many advanced therapy medicinal products (ATMPs) in development in the EU are for rare diseases and conditions. Since the...

Hydrogen Peroxide-Based Contact Lens Care Products: Consumer Labeling Recommendations - Premarket Notification (510(k)) Submissions
Draft Guidance for Industry and Food and Drug Administration Staff AUGUST 2022 FDA is issuing this draft guidance to provide labeling recommendations for hydrogen peroxide-based contact lens care...

FDA Issues Over-the-Counter Hearing Aids Final Rule and Guidance on Regulatory Requirements for Hearing Aid Devices and Personal Sound Amplification Products
Today, the U.S. Food and Drug Administration (FDA) issued a final rule: Establishing Over-the-Counter Hearing Aids to improve access to safe, effective, and affordable hearing aids for millions of...