Clinical Research Solutions
5 Ways the Medical Industry is Using Data Science
In the era of technological disruption, data science is a disruptor for the books. Today’s data scientists develop processes, algorithms, and systems to mine structured and unstructured data with the...
How to Prepare for Laboratory Partner Selection during CBD Product Development
The interest in developing consumer products or therapies derived from Cannabis or CBD is continuously growing. As these new products come to market, there is increasing need to comply with...
Clinical Research Solutions
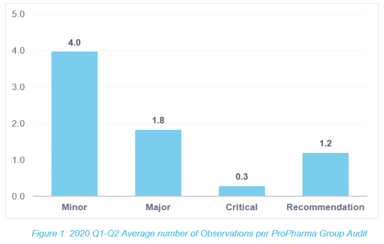
How Many Observations are Hiding in Your Quality System?
Reflecting on 2020, we have become accustomed to the global shift into a world of virtual, remote, restricted, and paused. One no longer flinches when hearing that something has been modified,...
Quality & Compliance
6 Tips to Prepare Your Medical Cannabis Facility for Inspection
You may be considering building a new facility for growing, harvesting, and processing medical cannabis, or perhaps you have an existing facility and want to export to the European Union. What should...
Regulatory Strategy in Pharma & Biotech Submissions Trial Design
Drug development can be a protracted and multifaceted process. This is often the case for startups and newer organizations, which may not have dedicated regulatory compliance departments and...
Clinical Research Solutions
Meet the Expert: Simona Mills, PMP
ProPharma has launched a “Meet the Expert” series introducing you to our experts from around the world. This series will help you get to know who we are, and how our colleagues work to improve...
Clinical Research Solutions
Why It's Smarter to Play the Long Game When Planning Your FDA Submission
The drug development process is long and expensive. You are doing yourself and your product a huge advantage if you enter and go through the process with a well-researched plan. When planning your...
6 Ways to Increase the Value of Your Cell and Gene Therapy or ATMP Development
As the field of modern medicine is changing, so should the development strategies of these new therapies such as cell and gene therapy (CAGT) products, also known as advanced therapy medicinal...

Clinical Research Solutions
How to Ensure Your Device, Software, and Drug Are All Ready for a Pre-Approval Inspection
When drug or device manufacturers apply for marketing approval of a new product, the FDA may conduct a pre-approval inspection (PAI). The PAI is performed to help the Agency assure that a...

Clinical Research Solutions
EMA vs. FDA Virtual GCP Auditing Guidance: What You Need to Know
In response to the COVID-19 pandemic, global regulatory authorities adopted a pragmatic virtual auditing approach. This approach includes the flexibility and procedural simplifications to maintain...
EMA Vaccine Applications for COVID-19 Explained
Preventive vaccines are a special kind of medicinal product, as they are administered before a disease instead of during. You are probably familiar with preventive vaccines from your own or your...

Regulatory Sciences
What You Need to Know About the ICH
The FDA has a strong interest in promoting the health and safety of U.S. citizens; in addition, it also supports the advancement of the global harmonization of regulatory requirements and practices....
Clinical Research Solutions
Virtual GCP Auditing: Your Questions Answered!
How quickly the auditing landscape has changed! Less than one year ago if ProPharma were asked to perform a clinical audit on your firm’s behalf, we would reply with "when, what, and where?" Today...
Clinical Research Solutions
Key Takeaways: New Draft Guidance on Cannabis and Clinical Research
On July 21st, 2020, the FDA released a draft guidance document for developers of cannabis and cannabis derived compounds, aptly titled “Cannabis and Cannabis Derived Compounds: Quality Considerations...
Quality & Compliance
7 “Aha Moments” in Data Integrity
If you are interested in data integrity, you likely have read many articles about the topic. This blog delivers 7 new insights to data integrity, looking at it from different angles. Seven ways to...
Clinical Research Solutions
4 Hidden Obstacles to Clinical Trial Success
It's no wonder scientists begin clinical trials with great excitement: combine promising discoveries with the best test sites, scientists, and as many qualified, willing patients as possible, and off...