Clinical Research Solutions
The Truth Can Hurt - But Hearing It at the Right Time Can Save Time and Money
No one has an ugly baby. At least, no one thinks their baby is ugly. Every new parent thinks their baby is the most beautiful baby of all time. But the unfortunate fact is that there are ugly babies....
Clinical Research Solutions
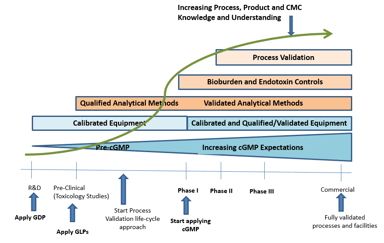
Is Your Technical Transfer Process Aligned with Process Validation Requirements?
There has been a lot of discussion recently concerning process validation and technology transfer, including utilizing virtual technology transfers to quickly move products through the development...
Clinical Research Solutions
5 Tips for Achieving Regulatory Success in 2021
As we begin to wrap up the year and look ahead to 2021, it is critical to prepare your team for what is coming in the weeks and months that lay ahead. Preparation and preparedness are key to ensuring...
Clinical Research Solutions
A Guide to the Clinical Study Report
What is a Clinical Study Report? A Clinical Study Report (CSR) is a document that describes the methods and results of a clinical study or trial, along with a short discussion of key findings related...
Clinical Research Solutions
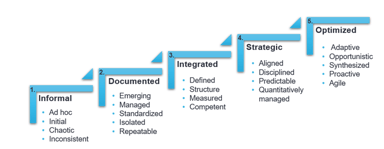
Improve Quality Using an Organizational Maturity Model
If this is your first introduction to an Organizational Maturity Model (OMM), you may have a few questions. What is an OMM? What are some common obstacles I might face when implementing an OMM? How...
Clinical Research Solutions
Understanding EMA and FDA Regulations on Nitrosamine Control
On September 26, 2019, the European Medicines Agency (EMA) released an advice to Marketing Authorization Holders (MAH) of human medicines to review their drug products on possible presence of...
Clinical Research Solutions
The Role of Clinical Data and Clinical Data Science
Clinical data and its analysis are critical to clinical research. Ensuring the overall quality of clinical data is then paramount to ensuring quality care and appropriate decision-making in the...
Clinical Research Solutions
Are Your Compliance Obligations Being Properly Upheld? Avoid This Common Outsourcing Mistake!
Over the past several decades, the traditional approach to drug development and manufacturing has expanded to include the outsourcing of a range of functions from product development and testing, to...
Quality & Compliance
Pharmaceutical Tech Transfer Best Practices - A Quality Perspective
About 15 years ago, I was a project management director responsible for moving monoclonal antibodies (MABs) from Phase III clinical to commercial manufacturing. I had the distinct pleasure of working...
Clinical Research Solutions
How to Sustain Compliance with EU MDR (2017/745) After the Deadline
The Medical Devices industry breathed a sigh of relief for the new European Medical Device Regulation (EU MDR), but the May 2021 deadline is now right around the corner. The transition from the...
Unlocking Success with Clinical Trial Safety Monitoring During a Pandemic
Earlier this year I wrote to you about US FDA March 2020 issuance of a new guidance for industry, Investigators, and Institutional Review Boards regarding the conduct of clinical trials during the...
Clinical Research Solutions
Why Drug Approvals Are Never Slam Dunks
It's the FDA, Not the NBA Aside from being a spectacular thing to watch, the slam dunk is the highest percentage shot a basketball player can take. The likelihood of getting the ball in the basket is...
Clinical Research Solutions
Filing Using Registry-Based Studies? EMA Issues Draft Guidance
Recruiting clinical studies in a niche disease area can be challenging, but disease registries can provide the solution. The European Medicines Agency (EMA) has issued draft guidance on how...

Clinical Research Solutions
Uncover Opportunities for Improvement with an Annual Product Review
The Annual Product Review (APR), also known as the Annual Product Quality Review (APQR), is required for marketed products in an FDA-regulated environment. You may ask, "Why would I want to perform...
Clinical Research Solutions
Is Your Pharmacovigilance Team Ready for Brexit?
As we approach the final months of 2020, the pharmaceutical world begins, once again, to focus its thoughts on the impact of Brexit, not least in the world of pharmacovigilance. Of course, the UK has...
Clinical Research Solutions
EudraLex Volume 4, Annex 1 Update: What You Need to Know
EudraLex Volume 4, Annex 1 provides guidance for the manufacturing of sterile medicinal products that are intended for the European market. It has been updated several times, with the latest revision...