
Clinical Research Solutions
Human Subject Protection Regulations: Differences Between HHS’ & FDA’s Clinical Trial Rules
On Friday, October 12th, FDA issued a guidance document entitled “Impact of Certain Provisions of the Revised Common Rule on FDA-Regulated Clinical Investigations.” The document aims to help...

Regulatory Sciences
CBER Provides Sponsors with Policies and Procedures Regarding INTERACT Meetings
On Monday, October 1, FDA’s Center for Biologics Evaluation and Research (CBER) issued a document outlining the policies and procedures for scheduling and conducting INitial Targeted Engagement for...

Good Review Management Principles & Practices, Part One: Fundamental Values
On Tuesday, September 25th, the FDA published a draft guidance containing recommendations on good review management principles and practices (GRMPs) for new drug applications (NDAs), Biologics...
Clinical Research Solutions
FDA Releases Draft Guidance on Benefit-Risk Determinations for Devices
On Thursday, September 6th, the FDA released a new draft guidance regarding benefit-risk determinations in medical device premarket approval applications (PMAs), De Novo requests, and humanitarian...
Regulatory Sciences
Generic Failure: Why so Few ANDAs Are Accepted by FDA on the First Pass
Generic drugs are immensely important to the U.S. healthcare system. These drugs account for 89% of the prescriptions dispensed in the United States. And, over the last decade, generic drugs have...
Clinical Research Solutions
Implementing a Risk-Based Supplier Management Program
According to recent FDA updates on the implementation of the Safety and Innovation Act (FDASIA), nearly 40 percent of finished drugs are being imported, and nearly 80 percent of active ingredients,...
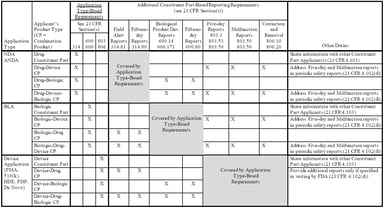
Clinical Research Solutions
Understanding the New Combination Product PMSR Guidance Documents and Impact on Industry
On March 20, 2018, the US Food and Drug Administration (FDA) released two new guidance documents to help companies comply with the December 20, 2016 final rule establishing postmarketing safety...
Clinical Research Solutions
FDA Steps up its Game on Generic Drugs: The Story Behind the Recent Focus on Generic Products
Throughout 2017, the FDA focused its attention on the regulation of generic drug products. In 2015, the Agency issued only two generic-related guidance documents. In 2016, there were seven. In 2017,...
Clinical Research Solutions
Transformational Leadership: Part II
Last week, I introduced a two-part series on transformation leadership. We defined transformational leadership, explored the concept, and discussed the benefits of adopting this approach. Well, now...

Clinical Research Solutions
Summary Considerations: Use of Electronic Records and Electronic Signatures in Clinical Investigations Under 21 CFR Part 11 – Questions and Answers
Here I provide some key summaries and considerations relative to FDA’s draft guidance that was submitted for review and comment in June 2017. If you don’t want to review the entire guidance, here are...
Clinical Research Solutions
Using a Matrix Approach to Media Fills in Sterile Compounding
A survey of FDA Form 483 observations issued to the 503B pharmacy industry reveals that outsourcing compounders are struggling to implement and manage compliant and risk-based approaches to aseptic...
Clinical Research Solutions
What Should Be on Your Clinical Trial Investigator Site Audit Checklist?
You live and operate in a regulated industry. Obviously, it’s crucial that you stay in compliance during your clinical trials. That’s because failure to do so has enormous and expensive consequences....
Clinical Research Solutions
How to Write an Effective Quality Investigation Report
In 2016, the FDA issued hundreds of 483 observations across the Drug and Device industries for failing to thoroughly review or investigate issues. This topic consistently hits the top five most...
Clinical Research Solutions
Should Data Integrity Detection be a Part of Routine cGMP Training Programs?
The FDA’s focus on data integrity in recent years has proven that it remains an industry issue. The focus has resulted in significantly increased issuance rates of 483 observations, warning letters,...

Clinical Research Solutions
Data Integrity 101: Is your data compliant?
Data integrity has recently been in the agency spotlight. In part because of the draft data integrity guidance issued April 2016, but primarily due to an increased number of inspection findings...
Clinical Research Solutions
Clinical Quality Systems & the Outsourced Model
The landscape of clinical trials is evolving. The changes that are happening are due to the increased number of FDA-regulated trials, as well as a rise in the complexity of clinical protocols. As...