
Regulatory Sciences
The Moving Regulatory Landscape for Gene Therapy Trials in EU
Submitting a GMO Application for a Clinical Trial with AAV Viral Vectors Navigating the regulatory landscape for clinical trials in the European Union (EU) has become more streamlined with the...

Regulatory Sciences
The Moving Regulatory Landscape for Gene Therapy Trials in EU: Part 2
Change in the Submission of the Summary Notification Information Format The Summary Notification Information Format Form As of January 31, 2023, Sponsors are required to submit a Clinical Trial...

Quality & Compliance
The Moving Regulatory Landscape for Gene Therapy Trials in EU: Part 1
The Benefit of the Common Application Form for GMO Applications GMO Application Not Included in Clinical Trial Regulation With the transition period for the Clinical Trial Regulation No 536/2014...

Quality & Compliance
5 Key Challenges in the Development of Gene Therapies
Over three decades of gene therapy (GT) clinical trials have yielded many innovative and promising treatments for pernicious genetic disorders but industry has shown slow development and dashed...

Medical Information
The Role of Patient Support Services in the Evolving World of Cell & Gene Therapies (CAGTs)
Cell and Gene Therapies (CAGTs) represent a transformative step in medicine, offering unprecedented potential to treat and potentially cure diseases once deemed untreatable. However, the path to...

Quality & Compliance
How to Decentralize Cell and Gene Therapy Treatments
The Challenge: CAGT Treatments Require Time and Lack Availability Persons diagnosed with life threatening diseases which are now treatable with cell and gene therapies (CAGT) often needed to wait for...

FDA Publishes Guidance for CAR T Products
In January 2024, the FDA Center for Biologics Evaluation and Research (CBER) Office of Therapeutic Products issued a guidance document- Considerations for the Development of Chimeric Antigen Receptor...

Quality & Compliance
Decentralization of Cell and Gene Therapy
Patients typically go through the same thought process when they have been diagnosed. After the relief of knowing that their ailment was correctly diagnosed, most patients want to know the path back...
Regulatory Sciences
How to Maximize the Value of Cell and Gene Therapy Products: 5 Tips for Success
Product development is an extremely time consuming and challenging process for any pharmaceutical, biotechnology, or medical device, but when it comes to cell and gene therapy products, the process...
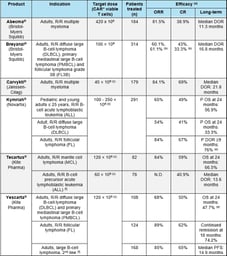
CAR-T Cells: Challenges, Lessons Learned, and Guidance for the Clinical Development
It comes as no surprise to any pharmaceutical or biotech company that planning the clinical development of CAR-T cells is an extremely challenging endeavor: high efficacy is expected in each targeted...

7 Critical Factors for Successful Selection of CDMO for Cell and Gene Therapy Manufacturing
Developing, optimizing, and manufacturing Advanced Therapy Medicinal Products (ATMP's), such as Cell and Gene Therapy (CAGT) products is extremely complex. The choice of a reliable Contract...
Quality & Compliance
Fundamentals of Data Integrity for ATMP Development
Advanced Therapy Medicinal Products (ATMPs), or Cell and Gene Therapies (CGT), have the incredible potential to cure devastating illnesses, such as cancer, on a more personalized level. But, due to...
Quality & Compliance
Deep Dive: FDA Guidance on Gene Therapy Products: RCR Testing and Monitoring
In January 2020, FDA published a guidance document entitled “Testing of Retroviral Vector-Based Human Gene Therapy Products for Replication Competent Retrovirus During Product Manufacture and Patient...
Quality & Compliance
5 Steps to CMO Identification and Selection for Cell and Gene Therapies
After assuring clinical validity, finding and managing the right contract manufacturing organizations (CMOs)/contract development manufacturing organizations (CDMOs) is a Sponsor's major concern when...
Quality & Compliance
Why is Process Optimization so Important in Cell and Gene Therapy Product Development?
A recent survey of experts from 145 cell and gene therapy (CAGT) companies revealed the ability to appropriately optimize the manufacturing process as their top concern. Red flags have been raised...
Quality & Compliance
The Importance of Using a Multidisciplinary Approach in Early-Stage Development for Cell and Gene Therapy Products
Implementing a multidisciplinary approach in cell and gene therapy product development is critical to the product’s eventual success or failure. In our experience, the consequence of not effectively...