
June 8, 2026
Modernizing Case Management for Global Compliance
Discover how E2B (R3) is modernizing pharmacovigilance case management, improving compliance, and streamlining adverse event reporting across global regulatory authorities.
ProPharma offers global Clinical Quality Assurance consulting to ensure inspection readiness and regulatory compliance across pharmaceutical, biologics, and medical device sectors. Our experienced QA professionals deliver end-to-end support—from GCP audits, SOP development, and CAPA management to trial oversight and mock inspections—tailored to your needs in every region. Wherever your clinical programs operate, we help you maintain quality, integrity, and confidence.

A strong Clinical Quality Assurance (CQA) framework is essential for maintaining the integrity, reliability, and compliance of clinical programs. Our CQA services provide proactive to reactive quality support to sponsors and CROs, ensuring adherence to regulatory standards and industry best practices. Ensure Clinical Quality & Compliance with Expert Support.

Oversight and compliance support for CROs, investigator sites, central labs, pharmacies, animal/toxicology studies, SaaS providers, and other vendors to ensure regulatory adherence and performance.
Identifying and resolving clinical trial issues to mitigate risks and improve study outcomes.
Thorough assessments of Trial Master Files to ensure completeness, accuracy, and regulatory compliance.
Tailored support to address specific clinical quality challenges and operational needs.
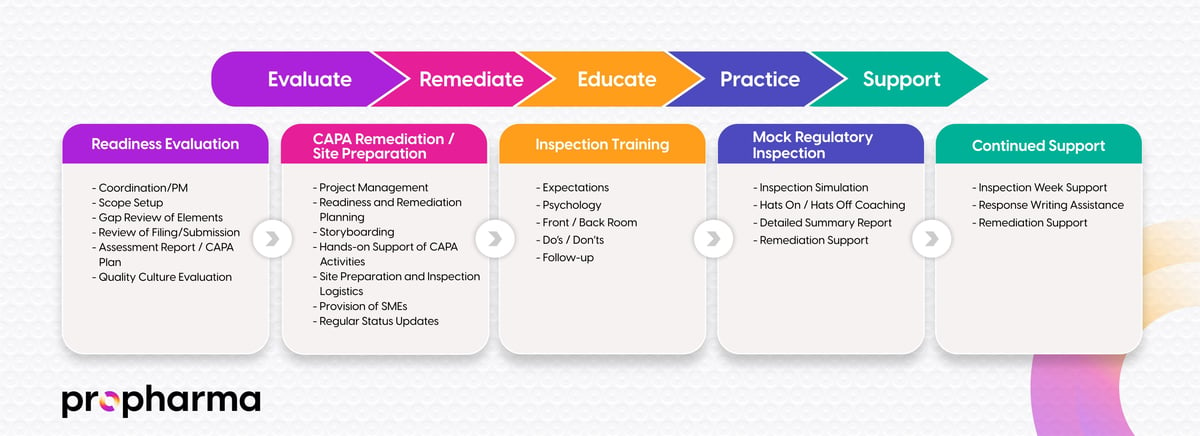
A structured auditing approach is essential for identifying compliance gaps, mitigating risks, and ensuring regulatory readiness. Our clinical auditing services help organizations maintain compliance with global regulatory expectations.
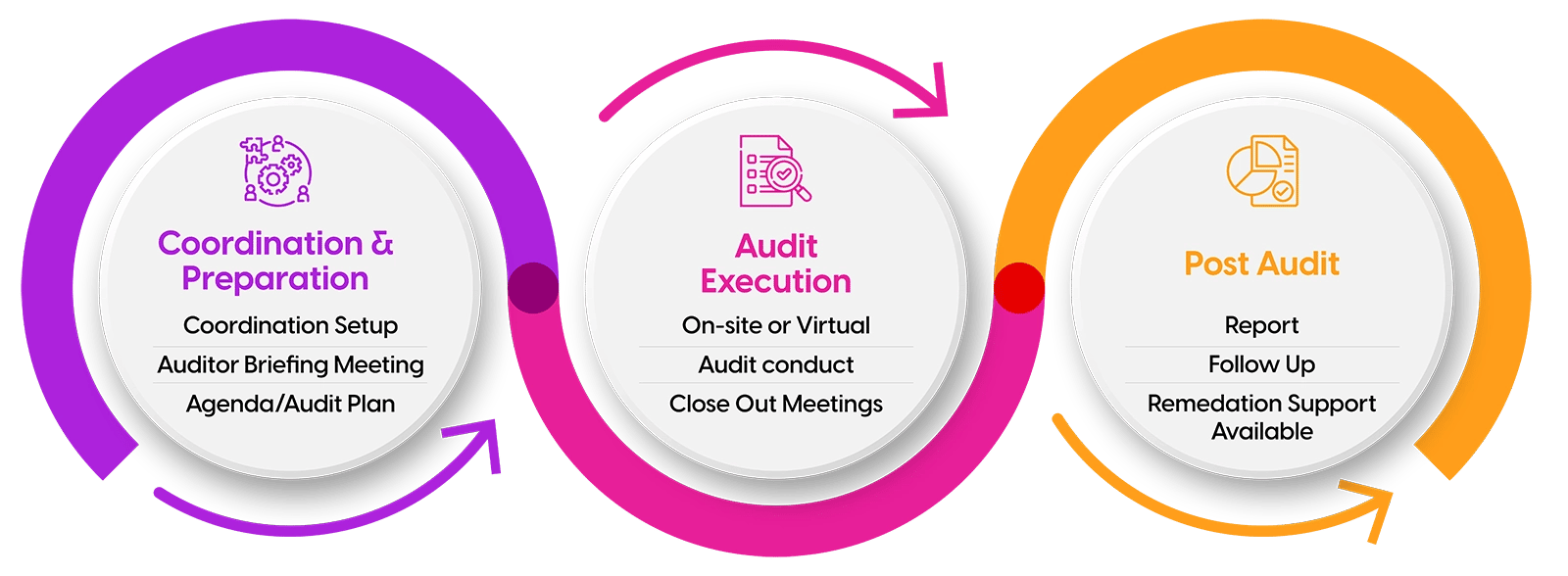
Regulatory inspections can be complex and high-stakes. Our inspection readiness services ensure your organization is fully prepared to meet global regulatory expectations with confidence.

A well-structured Clinical Sponsor Quality Management System (QMS) is essential for regulatory compliance, operational efficiency, and risk mitigation. At ProPharma, we help sponsors design, implement, and optimize QMS frameworks tailored to their clinical study and organizational needs.
The process begins with a kick-off meeting to ensure alignment with our client and key stakeholders to review unique requirements to consider.
Moving into the execution phase, we assess and implement QMS improvements:

Evaluating your current QMS and scaling solutions to fit your organization’s size, scope, and regulatory requirements.
Creating and refining policies, SOPs, and quality documents to ensure compliance with industry standards.
Supporting the seamless integration of quality management systems across your operations.
Providing in-depth training on QMS best practices, regulatory expectations, and continuous improvement strategies.

Whether you're building a new QMS or enhancing an existing system, our experts provide tailored solutions to ensure compliance and efficiency.
At ProPharma, we provide expert clinical consulting services to medical device and diagnostics companies. Helping to navigate and ensure compliance to complex global regulatory requirements while achieving operational excellence.
Our team specializes in supporting the unique requirements of medical device companies in meeting international compliance standards, including:

Partner with us for expert clinical compliance and quality solutions. Contact ProPharma today to learn how we can support your organization's Clinical Quality Assurance needs.

June 8, 2026
Discover how E2B (R3) is modernizing pharmacovigilance case management, improving compliance, and streamlining adverse event reporting across global regulatory authorities.

June 4, 2026
Learn how the UK's IRP can help FDA-approved therapies achieve faster UK market access, accelerate launch sequencing, and support global regulatory strategy.

May 27, 2026
ProPharma announces Adrian Pencak as Chief Commercial Officer, bringing 25 years of leadership experience to enhance global commercial capabilities and client partnerships.

January 27, 2026
ProPharma’s greenhouse gas reduction targets are validated by SBTi, aligning with the Net-Zero Standard and a commitment to reach net-zero by 2050.

January 31, 2025
ProPharma wins Silver at ECCCSA for AI innovation in Medical Information, enhancing efficiency and quality in delivering accurate medical information.

October 9, 2024
ProPharma wins CPHI Pharma Award for excellence in regulatory and compliance innovation, enhancing efficiency and accelerating market access for life-saving therapies.

June 10, 2026
As advanced therapy developers move from research-stage operations toward commercialization, they often face significant challenges in scaling quality systems, aligning regulatory strategy, and...

May 20, 2026
When patient enrollment rapidly exceeds expectations, clinical trials can face significant operational strain, putting data integrity, timelines, and critical sponsor decisions at risk. A mid-size...
.png?width=320&height=185&name=BLA%20Case%20Study%20-%20Clinical%20Hold%20to%20Approval%20(RS).png)
May 20, 2026
A critical CMC change during Phase 3 development led to a partial clinical hold, putting timelines, regulatory alignment, and commercial readiness at risk. In this case study, learn how we partnered...
.png?width=320&height=185&name=4%20-%20Quality%20%26%20Compliance%20(650%20x%20425%20px).png)
June 2, 2026
A compliant and efficient EU–UK GMP–GDP supply chain requires a clear understanding of roles, responsibilities, and operational integration across importation, certification, and distribution...
.png?width=320&height=185&name=Webinar%20Thumbnail%20Quality%20%26%20Compliance%20(650%20x%20425%20px).png)
April 23, 2026
As cell and gene therapies transition from clinical development to commercialization, organizations face evolving regulatory expectations, expanded CMC requirements, and increased MAH...

April 9, 2026
As European Medicines Agency Policy 0070 enters its expanded Step 2 phase, sponsors face increased document volumes, heightened transparency obligations, and greater scrutiny of anonymization and CCI...
.png?width=320&height=185&name=BLA%20Case%20Study%20-%20Clinical%20Hold%20to%20Approval%20(RS).png)
May 20, 2026
A critical CMC change during Phase 3 development led to a partial clinical hold, putting timelines, regulatory alignment, and commercial readiness at risk. In this case study, learn how we partnered...

April 9, 2026
As European Medicines Agency Policy 0070 enters its expanded Step 2 phase, sponsors face increased document volumes, heightened transparency obligations, and greater scrutiny of anonymization and CCI...