
Pharmacovigilance
The LPPV Network: An Essential Guide to Local Person Responsible for Pharmacovigilance
Every marketing authorization holder (MAH) needs a pharmacovigilance system to guarantee the safety of its products. Within the EU a pharmacovigilance system is defined as a system used by an...

Pharmacovigilance
The QPPV: An Essential Guide to the Qualified Person Responsible for Pharmacovigilance
When submitting marketing authorisation applications (MAAs) in Europe, the applicant (Marketing Authorization Holder, MAH) shall already have set up a pharmacovigilance system. A Summary of the...
Regulatory Sciences
Early engagement with Health Technology Assessment authorities will accelerate product launch and improve chances for reimbursement
Pharmaceutical companies should understand EU Health Technology Assessment (HTA) authorities requirements early in the product development phase. Engagement with HTA authorities during clinical...

Regulatory Sciences
5 Benefits of Receiving EU PRIME Designation for Medicine Developers
What it is, why it matters, how you can apply and how we can help. What is the PRIME Scheme? You might be forgiven if you don’t know what the PRIority MEdicines (PRIME) scheme is; but if you are in...
Maximising on Scientific Advice Procedures in Europe
A unique opportunity to interact with medicine regulators in Europe Are you considering requesting scientific advice in Europe? We can help you navigate the various procedures within the European...
EMA adopts first list of critical medicines for COVID-19
News On 7 June 2022, EMA's Medicines Shortages Steering Group (MSSG) adopted the list of critical medicines for the COVID-19 public health emergency. The medicines included in the list are authorised...
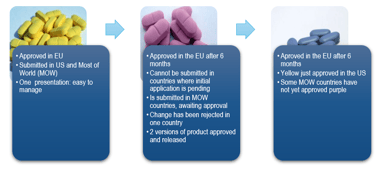
CMC Regulatory Dossier Compliance: A GMP Requirement
Maintaining compliance in the dynamic regulatory Chemistry, Manufacturing and Controls (CMC field can be quite a challenge. A CMC regulatory dossier compliance assessment is a critical component and...
Regulatory Strategy for Clinical Trials in the European Union
Setting up a clinical trial in the European Union (EU) has historically been an expensive and time-consuming business. With 27 individual member states each requiring its own review and approval, it...
