After panning over the Boston Business Journal and seeing yet another local VC (Venture Capital) firm raise a record $350 million in venture capital for Massachusetts life science, I asked myself: “how sustainable can this get?” The ability to raise money like this in the era of the post-COVID pandemic is a real achievement for the field, but suiting investors to increase operational value is one thing, and commercialization is a whole other animal.
Big Pharma increasingly places more and more of its bets on Massachusetts (MA) Biotech firms (that are not even out of what the FDA terms as "proof of concept") demonstrate a genuine willingness to partner early on in development to obtain a much-needed pipeline lacking innovation.
The same indications that Pharma has served over a century are no longer feasible as they have primarily addressed disease symptomatically. Thus, the idea of using cellular machinery from humans, animals, and even viruses to address the root cause of a human ailment has become a real game-changer.
The international regulatory term for such a technology is Advanced Therapeutic Medicinal Products (ATMP), which encompasses cell and gene therapy technologies. ATMPs are being developed for highly prevalent diseases and rare orphan drugs with no other treatments.
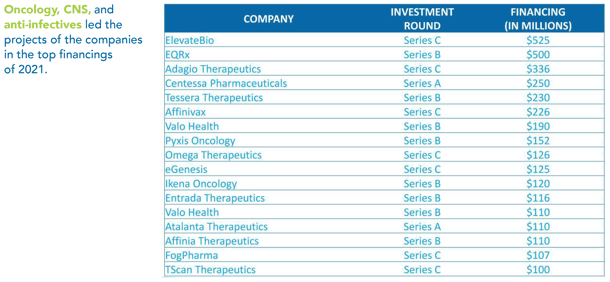
ATMP Funding in Massachusetts
The pace of funding for MA-Biotech has increased exponentially. According to the 2021 snapshot report from Massachusetts Biotechnology Council, Massachusetts-headquartered companies raised $4.3 billion in venture capital funding in the first quarter of 2021 alone, after raising $5.8 billion in all of 2020.
This funding rate will continue to be a viable opportunity for the life science consulting field to serve this growing economy, not to mention the world's best sports town.
Chasing a Cheetah
Notably in the case of ATMPs. As an auditor trying to assess compliance to an industry that I feel is still in adolescence, this sector of Biotech holds many challenges for sponsors and Life Science service providers alike.
Boston Cambridge Biotechnology space has grown so fast throughout my career that it has forced my colleagues and me to find new ways and opportunities to keep up with its pace.
Now more than ever, it is vital that we learn as the industry learns, even if it feels like we are chasing a cheetah some days.
However, this genetic juggernaut evolving and revolutionizing modern medicine is reaching a watershed moment in its ability to sustain its growth.
ATMP Challenges - Space
The high demand for lab space and an ever-increasing competitive job market requiring skilled labor have presented logistical challenges for ATMPs. In addition, the decline in lab space available to sponsors has forced MA biotech companies to move out of its nucleus of Boston-Cambridge, which has sparked clusters and incubators all over the state.
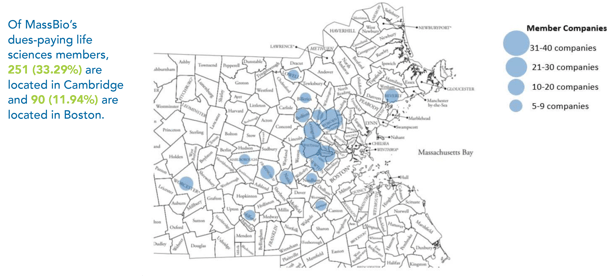
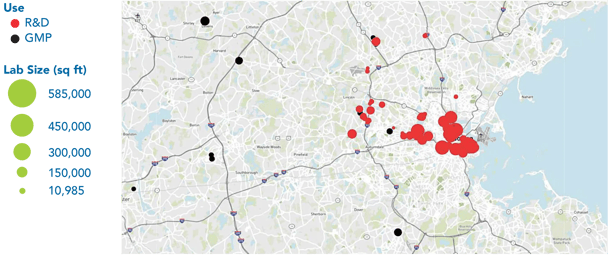
Now that we are almost "back to the norm" from COVID, firms still wary of infection transmission rates are increasingly going to remote work. This fear may or may not free up lab space, but it certainly will make more offices in need of new tenants available.
ATMP Challenges - Supply Chain
Another genuine concern for Massachusetts and the global ATMP sector is the ability to procure and maintain a steady supply of high-quality Viral vectors, Plasmids, and critical starting materials. ATMP supply chain is now being circumvented by three factors causing a high demand for these materials.
Firstly, Cellular Therapies require "Vectors" to alter the genetic code of cell receptors in vitro and then administer them back to the same patient. These cells are considered Autologous cells as they are obtained, processed, and transfused into the same individual. Autologous: Auto means self. Typical cells used for this approach are CD4+ helper T cells and CD8+ cytotoxic T cells.
This approach is called immunotherapy or immuno-oncology, as many cell therapies globally are being developed for specific cancers.
How CAR T-Cell Therapy Works
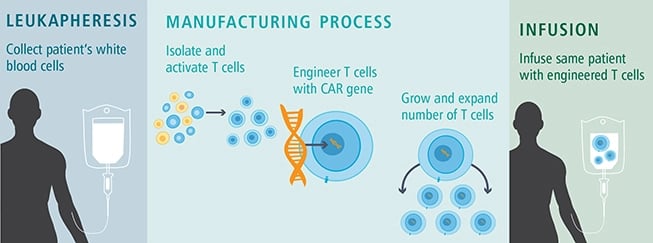
An example of an FDA-approved Cellular Therapy for infusion is Abecma® (idecabtagene vicleucel). Abecma is the first cell-based gene therapy approved by the FDA for treating multiple myeloma. Abecma cell therapy was developed and commercialized in the U.S. as part of a co-development, co-promotion, and profit share agreement with Bristol Myers Squibb and Bluebird bio.
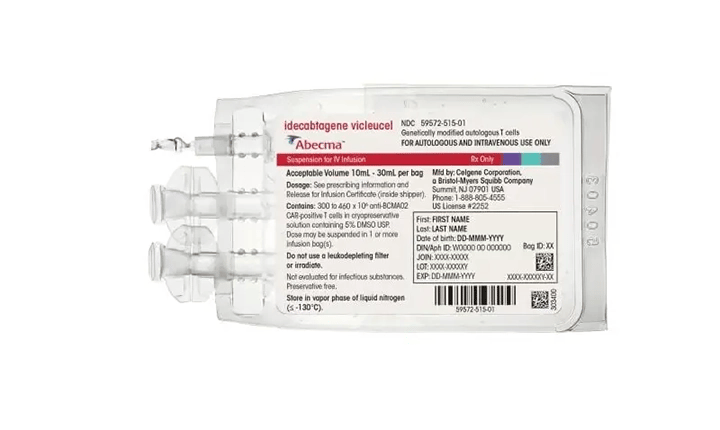
The second factor for high demand, gene therapy products (GTP), is the use of the same "vectors" used in cell therapies. Instead of delivering genetic information into cells outside and put back into the human body, gene therapy utilizes the vector to provide pharmacological effect to specific organ tissue cells like hepatocytes found in the liver. An example of an FDA-approved GTP is Zolgensma®, and its active ingredient is (onasemnogene abeparvovec-xioi).
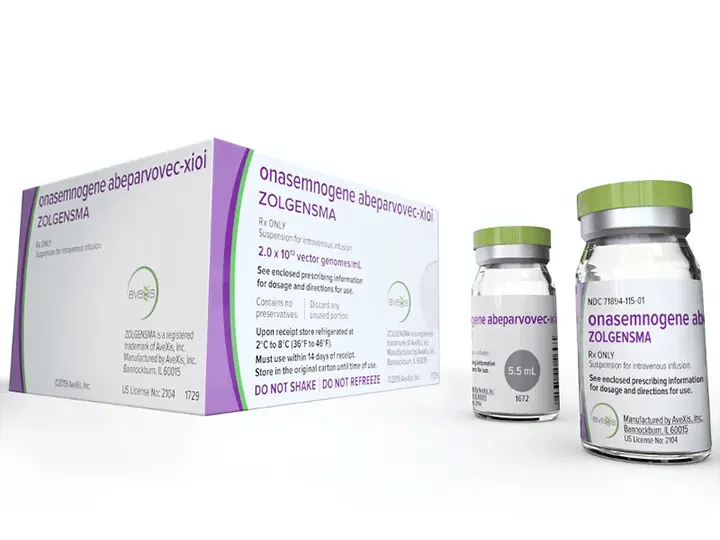
As GTP are large molecules, they cannot withstand terminal sterilization in final form and, in most cases, need Aseptic processing as a liquid vial. The safest and most stable way of administration for an injectable is to have it manufactured as lyophilized cake, as it is much more stable in this form.
Advances are coming in GTP formulation via liposomes and cellular cargo exosomes, allowing for a more durable product that can be resuspended in saline. The mode of action for the GTP is the scene here below.
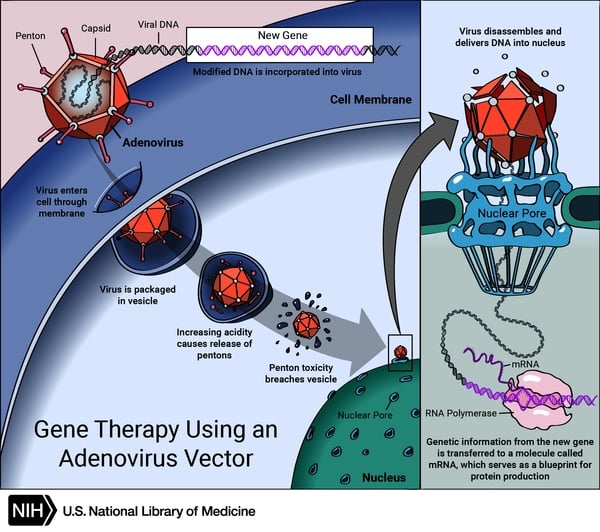
There are four main viral vector types (adeno-associated, adenoviral, lentiviral, and retroviral), each with unique characteristics, uses, and limitations.
A third factor for the high demand is the application of viral vectors in vaccinology (particularly for the COVID pandemic of 2020). A growing level of information supports recombinant viral vector usage as a means of vaccination. Studies have demonstrated that when antigen-expressing viral vectors are used, they elicit T- and B-cell responses that are wider and of a more significant order of magnitude than after DNA immunization alone.
COVID Pandemic has brought supplies and importation to a standstill. Many of my close colleagues are finding it even more difficult to obtain 50ml polystyrene tubes! Biotech is not the only industry to feel this pain. Next time you go to Staples to purchase your ink cartridge, the likelihood of it being in stock has diminished considerably, and the Suez Canal debacle that took 4 months to get resolved hasn't helped matters.
These three modalities for ATMP all utilize customized Vectors that require independent manufacturing and controls to be efficacious. They are considered critical raw materials in ATMP for both cell and gene therapy applications.
Meeting the Challenge
To meet demand and produce safe and effective ATMP, contract Vector manufacturers need a qualification audit conducted by the sponsor or third party to ensure they are in a state of control for Good Manufacturing Practice (cGMP) regulations. This is typical expectations for a clinical manufacturing setting.
In summary, we can now see how broad the Boston biotech space is, with multiple modalities and far-reaching healthcare implications. Further, we know the growth rate and its growing pains in an environment where the regulatory landscape is ever-changing and relies on the scientific community's best judgment that aims to balance technological innovation with patient safety.
ProPharma is strategically positioned to become an active partner in this diverse ATMP economy. Engagement with sponsors to understand their needs is vital and allows ProPharma to tailor its business solutions to deliver value.
If you are interested in learning more about ProPharma, contact us today.

