There are many ways in which you can evaluate a Contract Manufacturing Organization (CMO) to identify the best fit for your company's needs.
Every company knows timing is critical to success. But even the most well-designed products can be doomed by poor timing. Have you been tasked with leading the CMO selection process for your company? We’ve been in your shoes and understand just how critical this step is for future growth and success. Choosing the wrong contract manufacturer can be disastrous, leading to delays in supplies or even jeopardizing regulatory approval.
A good selection process, with realistic selection criteria, is essential when choosing the right contract manufacturing partner. To help you with this process, ProPharma has developed the CMO Compass® tool. Developed by our industry experts, and kept up to date with continuous data collection, this tool takes a systematic approach to the selection process.
No two CMOs are the same – they all have different strengths and weaknesses. Based on a series of questions, the CMO Compass serves as a guide to help match companies with the right CMO. It's quick and easy to use and can save you precious time that you could spend on the many other tasks you have on your plate.
Considerations When Selecting a CMO
The radar graph presented below shows the different qualities that are considered during our selection process. For example, a CMO/CDMO with strong compliance management or good audit history can help avoid submission rejections in later stages.
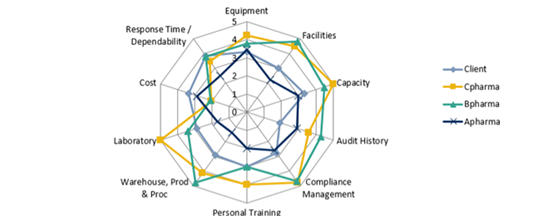
It is critical for companies to know and understand the steps required in the selection process. With that said, we advise clients to begin with the Target Product Profile (TPP) and perform the following 5 Steps to CMO Identification:
- Map out the transfer process, including applicable boundaries and conditions
- Set expectations with key stakeholders
- Identify CMO/CDMOs to be surveyed
- Provide the process map and list of expectations to each CMO/CDMO being considered
- Create a detailed report
Based on the results generated by the CMO Compass tool, clients are provided with their top CMOs/CDMOs. Next, the CMO/CDMOs will be screened based on selection criteria. The initial selection criteria serve as a guide but can be adjusted throughout the selection process if needed.
For instance, equipment and lab facilities may be the initial criteria; however, additional criteria such as auditing history and training may become more important and be added to the selection criteria later in the process.
The next step is to "narrow the field." Select 3-5 potential vendors to be included in the RFP process. Once the CMO/CDMOs list are narrowed down, ProPharma will document the decision and perform audits on your behalf. Although audits are a part of our complete quality system services, we can also work with you on an audit-only basis. The audit can be tailor-made based on your specific needs, and together we will plan your audit project. We will train the auditor specifically to your needs. No matter the location or type of audit, we charge a fixed price per On-Site-Audit-Day, allowing you to control the budget.
When the audit is completed, you will receive an audit report with all findings triaged as minor, major, or critical. Once all the shareholders have completely recognized and understood the concerns, we can start tackling them.
Next Steps
The next step is to have a thorough plan for technology transfer and/or scale-up. Technology transfers are a crucial step in the product development process, and excellence in pharmaceutical technical transfers is a “must have” for organizations today.
ProPharma uses a Nine-Gate Transfer Process to ensure that each stage is completed according to agreed constraints and allows leadership to quickly assess progress utilizing an industry-approved best practice. Transfer teams are guided by trained transfer professionals who understand how to maintain quality while triple constraints (Scope, Schedule, Cost) are fixed.
Our support in Tech Transfers can be as broad as overall Project Management, with a fully multidisciplinary team completing all activities until regulatory approvals or support from specific experts is needed within the Tech Transfer Teams. With more than 400 successfully performed technology transfers to various customers, our experts have created a guide to set your project up for success. Contact us to learn more.
This blog post has been updated since its original published date: November 23, 2021
TAGS: Quality & Compliance Contract Manufacturing Organization (CMO) Life Science Consulting CMO Compass

