Over the past several decades, the traditional approach to drug development and manufacturing has expanded to include the outsourcing of a range of functions from product development and testing, to manufacturing. As a part of their long-term strategic view, successful Sponsors understand the need to leverage the knowledge and resources that external specialist organizations bring to the table. Many times, the required resources do not exist in the Sponsor’s organization.
Sponsors of every size and type outsource clinical functions to contract research organizations (CROs) and manufacturing functions to contract manufacturing organizations (CMOs).
Virtual companies, run by a small team, outsource functions because they lack the financial or personnel resources needed to conduct clinical trials and manufacture their drug. Mid-sized companies may outsource so that they can focus on their core competencies while leveraging the niche expertise of contractors. Finally, large companies might decide that it is more effective for them to outsource functions because doing so will result in greater efficiency, shorter time to market, and lower costs.
While these are all valid business decisions, outsourcing is not a means of mitigating risks or transferring compliance obligations to contracted facilities. When it comes to compliance with FDA regulations, the buck stops with the Sponsor. Regardless of the relationship, outsourcing functions does not mean that a Sponsor has outsourced compliance obligations. It is understood that the Sponsor is responsible for the CRO’s or CMO’s quality.
Consequently, Sponsors must do everything within their power to ensure that their actions, as well as the actions of the CRO or CMO, are compliant, and it starts with having a quality system in place.
The Importance of a Quality System
Sponsors are responsible for making sure that every task associated with the clinical trials, manufacturing, and distribution of their drug is performed in compliance with FDA regulations. This is achieved by operating with a robust quality system designed to keep the Sponsor and its contracted facilities compliant.
The stronger the Sponsor’s clinical quality system, the more likely their study is to be compliant with Good Clinical Practices (GCPs) and able to quickly progress to the next phase in development. This helps the Sponsor save time and money when working with their CRO to remedy something uncovered by an internal, third-party, or FDA audit.
As for Good Manufacturing Practices (GMPs), a strong quality system provides for the proper design, monitoring, and control of manufacturing processes and facilities. This ensures that drug products meet the FDA’s quality standards.
No matter how well-designed a Sponsor’s quality system is when they are outsourcing functions, it’s crucial that they have a quality agreement that extends the Sponsor’s quality system to the contracted facility.
The Necessity of a Quality Agreement
As part of a larger outsourcing management plan, a Sponsor should have a comprehensive, written quality agreement with each of their contracted facilities. This agreement is a part of the Sponsor’s quality system and delineates what is expected of both parties by defining the obligations and responsibilities of the quality units of both parties.
Although there is no statutory or regulatory requirement for a quality agreement, regulations do require that quality unit responsibilities and procedures be in writing (21 CFR 211.22(d)). Despite the lack of a legal requirement, in their guidance on the matter, the FDA recommends that Sponsors and contractors operate under a quality agreement. Furthermore, the Agency’s guidance also states that inspectors will seek to review quality agreements during facility inspections.
Again, even though there may be a document that clearly sets out the obligations and responsibilities of the Sponsor and the contracted facility, that document does not have the power to transfer compliance obligations from the Sponsor to the contracted facility. If there is a failure in compliance on behalf of the contracted facility, the Sponsor will always be responsible.
The Key Criteria for Selecting the Right Compliance Partner
Regardless of the Sponsor’s size or their arrangement with a contracted facility, the Sponsor cannot outsource their compliance obligations. Because of this reality, they must keep their obligations in mind as they work with their contracted facilities.
ProPharma works closely with Sponsors to create a customized quality system design that promotes GCP and GMP compliance while accommodating each Sponsor’s unique circumstances. We start by conducting a gap assessment to determine the strength of their existing quality system and submit a report that details what we found and includes recommendations based on these findings.
What happens if you don’t have a Quality Management System (QMS)? No SOPs? No problem. Many of our clients are Clinical Phase 1 or Phase 2. ProPharma can build your QMS from scratch and make it “phase appropriate”. Depending on where your firm is at on the GCP or full GMP continuum, we build only what you need, when you need it. See the graphic below to better understand what “phase appropriate” looks like:
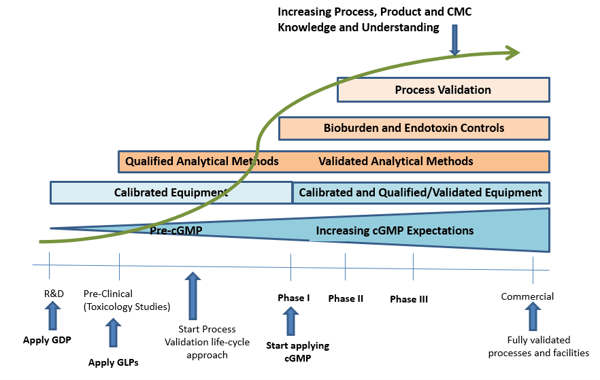
Based on a Sponsor’s needs, our team can build out as little or as much of the quality system as requested. When remediation is suggested, we can also help with related project management. From standard operating procedure (SOP) writing to building a quality framework to providing a solid methodology for GCP and GMP audits, our team helps Sponsors mitigate the risk of noncompliance.
To learn more about our expert services, contact us today.