North America
EMA
August 22, 2022
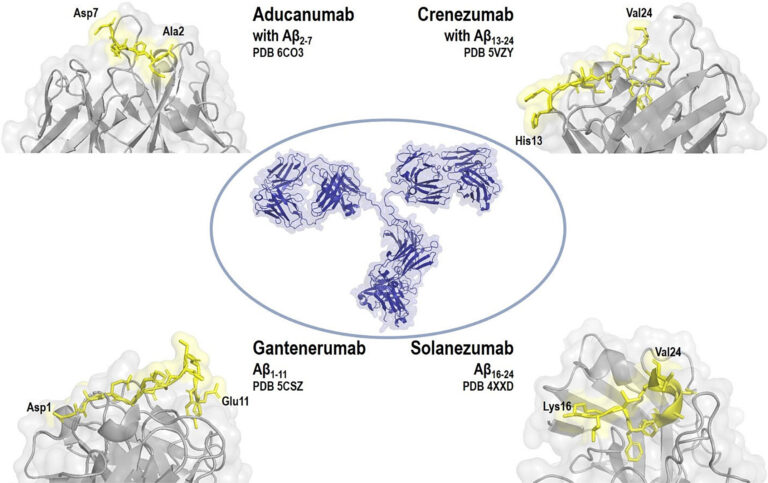
Orphan Designation of ATMPs for Rare Diseases: MPS II Case Study
Orphan Designation of ATMPs for Rare Diseases: MPS II Case Study Many advanced therapy medicinal products (ATMPs) in development in the EU are for rare diseases and conditions. Since the...
FDA
MHRA
August 8, 2022
Early engagement with Health Technology Assessment authorities will accelerate product launch and improve chances for reimbursement
Pharmaceutical companies should understand EU Health Technology Assessment (HTA) authorities requirements early in the product development phase. Engagement with HTA authorities during clinical...
MHRA
North America

August 1, 2022
UK Paediatric Investigational Plans – what do you need to know?? …and how is it all working in practice??
If a marketing authorisation is planned to be submitted in England, Scotland, and Wales (GB), an MHRA-approved paediatric investigational plan (PIP) is required. Up until January 1, 2021, PIPs were...
Europe
ILAP
![]()
July 25, 2022
How to Fast-Track medicine approval in the UK with the MHRA’s Innovative Licensing and Access Pathway (ILAP)
What is ILAP? What benefits does ILAP provide? How do you access it? With the dust of Brexit settling, the question on most people’s lips (well, those of us in the healthcare sector anyway!) was:...
FDA
North America
July 1, 2022
Clinical Pharmacology Considerations for Oligonucleotides
For the first time, the FDA has issued a draft guidance for industry on “Clinical Pharmacology Considerations for the Development of Oligonucleotide Therapeutics”. Oligonucleotides are short single...
ANDA
FDA

June 27, 2022
How to Interpret FDA’s Final Guidance - “Assessing the Effects of Food on Drugs in INDs and NDAs - Clinical Pharmacology Considerations”
On June 23, 2022, The FDA issued the final version of its Guidance for Industry titled “Assessing the Effects of Food on Drugs in INDs and NDAs - Clinical Pharmacology Considerations”. The most...
FDA
EMA

June 22, 2022
Maximising on Scientific Advice Procedures in Europe
A unique opportunity to interact with medicine regulators in Europe Are you considering requesting scientific advice in Europe? We can help you navigate the various procedures within the European...
Blog
General Regulatory

January 9, 2019
Year in Review: Taking a Look at Pharma's Top News Stories & Events from 2018
From FDA's approval of the first cannabis-based product in the U.S., to the classification of two Apple Watch apps and changes in the Agency's submission requirements related to Sponsor meetings,...
Blog
General Regulatory
![]()
May 21, 2012
Extended Review Timeline for Certain Applications under Proposed PDUFA V
The Prescription Drug User Fee Act (PDUFA) is due to be reauthorized by the end of September this year. Along with the usual increases in fees, the latest version of the act (PDUFA V) includes some...







